ITHACA, U.S.A. The Minglin Ma Lab in the Department of Biological and Environmental Engineering at Cornell University attacks some of the largest health issues, but in a very small way: using cutting-edge techniques in nanotechnology to develop potential treatments for diseases and other clinical problems. Their research primarily focuses on the use of nanobiomaterials, materials on the nanoscale level that are safe to be used with the body, in applications such as tissue regeneration and enhanced drug delivery systems.
Tissue Regeneration
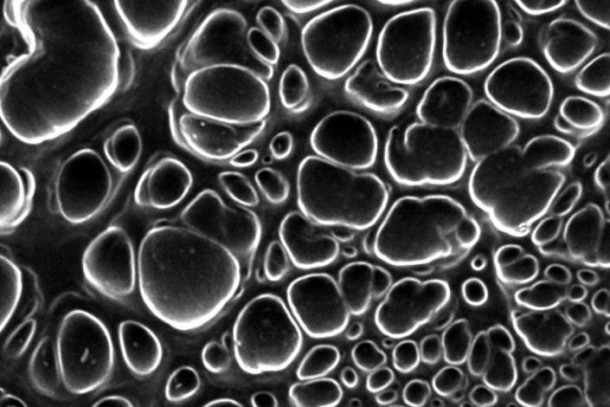
One of the most prominent uses of nanobiomaterials is in the field of tissue engineering, which focuses on the repair and regeneration of tissues in the human body. These materials are used to fabricate scaffolds, surfaces that allow cells to attach and proliferate, which eventually serve as the foundation for new, artificial tissue. Scaffolds can be fabricated in a variety of shapes and sizes and made using natural or synthetic materials, or as a combination of the two.
Polymers, chains of repeating chemical units, like polyesters in clothing or collagen in tendons, serve as the building blocks of these scaffolds. Scaffolds are typically constructed as three-dimensional (3D) structures, to mimic the real environment of cells within the body. One technique, known as electrospinning, pulls these polymers into strings, only nanometers in diameter, and collects them together in such a way that forms 3D surfaces resembling spider webs or fine tissue paper. Once the scaffold is formed, cells can be seeded onto them, which then grow and spread throughout the matrix of polymers to form a realistic, tissue-like structure.
This technique has been employed in the lab to develop artificial cartilage constructs to replace damaged cartilage in human joints. Because cartilage is an avascular tissue, meaning it does not receive blood flow, it has difficulty healing on its own, and so any damage to it is typically permanent and only continues to degrade from its onset, such as seen in patients with osteoarthritis (OA). Tissue-engineered constructs of cartilage cells embedded in scaffolds can be used to theoretically transplant this damaged tissue, providing a more successful alternative to conventional techniques of surgery.
Cell Encapsulation
Another area of major research in the Ma Lab is the use of nanoscale polymer scaffolds in the field of cell encapsulation and drug delivery. Cell encapsulation is a technique in which cells that secrete certain proteins, biological molecules that are absolutely crucial for proper function of the body, are trapped inside a 3D scaffold and injected into the body to release proteins that the body may be lacking. The scaffold primarily serves to protect the foreign cells from being attacked by antibodies that are released by the body’s autoimmune response, much in the same way a shark cage is used to protect the people inside from being attacked by sharks.
One of the most popular areas for this technology to be applied is in the treatment of Type I Diabetes, which is characterized by a deficiency in the protein insulin. Islets, pancreatic cells that secrete insulin, can be encapsulated in a polymer scaffold and injected into the body to provide the insulin the body needs. The cells would be able to release the exact amount of insulin needed in real-time, as opposed to current treatments that provide fixed doses, which may be too little or too much, only after the decrease in insulin has been detected by the person. In addition, this treatment would eliminate the need to prick fingers to test insulin levels in the blood throughout the day.
The integration of nanotechnology with the biomedical sciences is a rapidly expanding field providing new and innovative solutions to many of today’s healthcare issues, improving medicine from the nanoscale up.